What is the FDA’s Bioanalytical Method Validation Guidance for Industry?
Imagine looking at your car’s speedometer and seeing 60 miles per hour when you are really going 80. In healthcare, that kind of hidden error can be dangerous. Doctors rely on blood tests to check whether a drug is working, but a lab test is not perfect on its own. If scientists cannot measure how much of a drug is in your bloodstream, doctors cannot know whether a dose is safe or harmful.
To avoid those errors, labs have to prove that their measuring tools work. According to industry safety experts, each new testing method needs strict calibration before it ever touches a patient sample. This process, called method validation, is a stress test for the lab method. It checks that the background noise in human blood does not fool the system into giving false numbers.
Government watchdogs do not leave this to guesswork. Under the rules in the fda bioanalytical method validation guidance 2018, facilities must document proof that their tests are precise and reliable. That is what turns a chemistry experiment into a medical tool people can trust.

From Recipe to Result: Why Scientists Must Prove Their Lab Recipes Actually Work
Making the right cookie takes trial and error before a bakery can use the recipe. Labs work in a similar way when they build a test to measure a new drug. Creating the test is one job. Proving that it works well enough for patient care is another.
That first step is called method development. Chemists test ways to isolate the drug from a biological sample. But a method that works only some of the time is no good in healthcare. To keep patients safe, scientists move through clear method development vs method validation steps. Development is the build phase. Validation is the proof phase.
Once a method passes, the instructions are fixed in a Standard Operating Procedure, or SOP. Standard operating procedures for assay verification work like lab rulebooks. The goal is consistency. These documents make sure a scientist in Tokyo can follow the same method and get the same result as someone in New York.
Even with a strong rulebook, biology is messy. Blood is full of signals that can hide the drug. To make a method work with real patient samples, scientists have to cut through that noise and find one drug molecule among blood cells, proteins, and fats.

Filtering Out the Noise: How Labs Find One Drug Molecule in a Sea of Blood Cells
Finding a drug in pure water is simple. Finding that same drug in human blood is much harder. Scientists call that background interference the “matrix effect.” If the method is not calibrated well, a severe matrix effect impact on assay sensitivity can bury the drug signal and lead to the wrong dosage reading.
Rigorous analytical validation matters because the sample type, or matrix, changes the challenge:
- Pure Water: Clear and simple. There is little to interfere with the measurement.
- Human Blood: Crowded and complex. Proteins, fats, and blood cells can hide the drug.
- Solid Tissue: Even harder. Samples like liver or muscle must first be broken down into liquid before testing can start.
To work through that, tests need specificity and selectivity. Specificity means the test can identify the exact drug. Selectivity means it can find that drug in a messy sample without getting fooled by similar signals. In chromatography, which labs use to separate mixed liquids, selectivity and specificity help make sure the method measures only the analyte, the drug being tracked. Once scientists can see the drug clearly, they still have to prove the numbers land where they should.
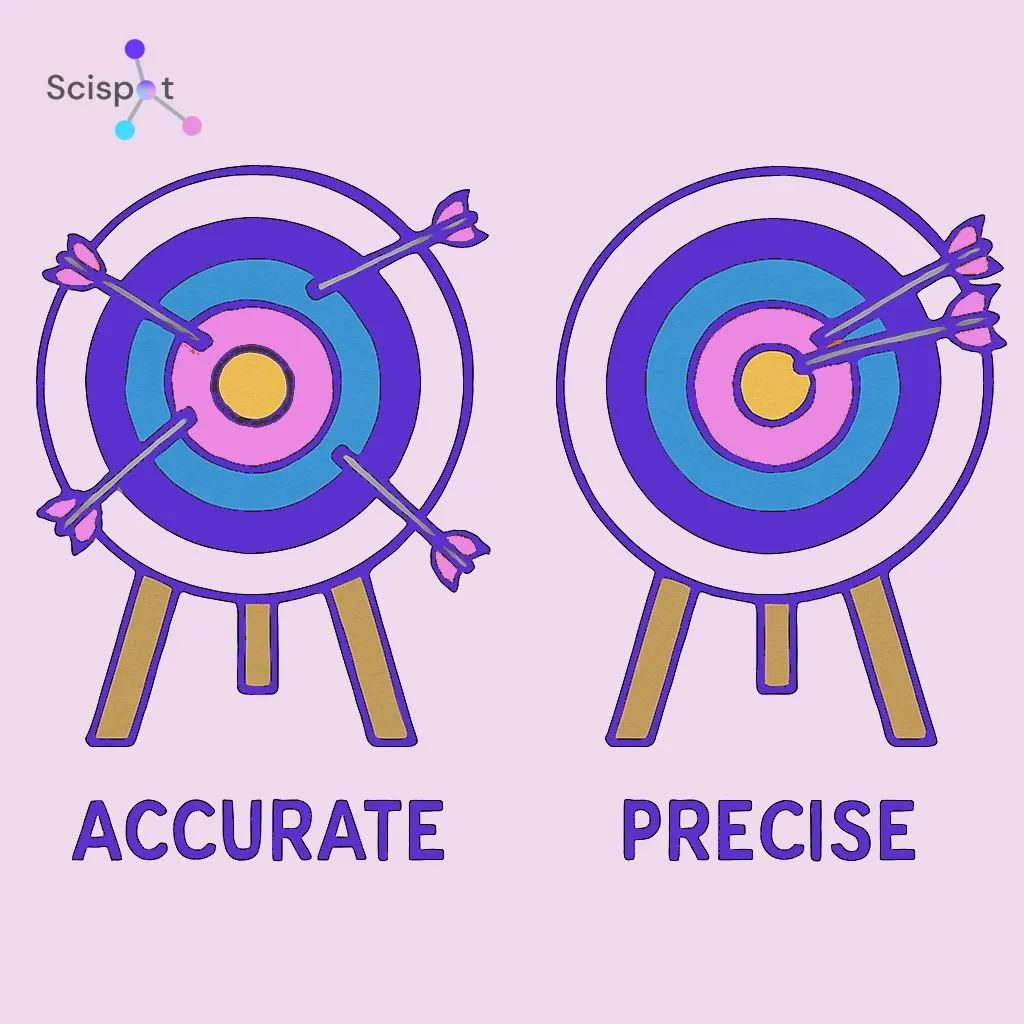
The Archery of Medicine: Why Lab Results Must Be Both Accurate and Reliable Every Time
An archer is a simple way to picture this. Accuracy means hitting the bullseye, or getting the true answer. Precision means the arrows land close together each time, even if they miss the center. A medical test needs both. It has to hit the right answer and keep hitting it every time.
Because patient safety is on the line, regulators set limits on how far results can drift. Under the fda bioanalytical method validation guidance 2018 lc-ms ms, measurements generally cannot be off by more than 15%. That 15% rule shapes the accuracy and precision requirements for small molecules, which includes many common drugs such as blood pressure medicine. A wider error margin could change a safe dose into a harmful one.
One good run is not enough. Scientific trust depends on reproducibility. A validated analytical method, meaning the lab’s step-by-step testing procedure, has to give the same result in New York on Monday and in Tokyo on Friday. That consistency supports clinical trials and the decisions built on them. Once the method proves it can hold steady, the next question is how little drug it can still measure with confidence.
Hearing the Whisper: Detecting the Smallest Trace of Medicine Without Losing the Signal
Measuring tiny drug levels in blood takes a sensitive method. No machine can measure an endlessly small amount. At some point, the background noise takes over. The point where the signal can still be measured with confidence is the lower limit of quantification, or LLOQ. It is the lowest drug amount the lab can measure without guessing.
To measure those tiny amounts, scientists use a calibration curve. It works like a paint chart at a hardware store. You compare an unknown sample against known reference points to figure out what matches. Labs do the same thing with known drug concentrations. Strict calibration curve range selection criteria make sure this mathematical ruler covers the drug levels expected in the body, so the results stay reliable.
Even then, a good machine cannot save a bad sample. If the blood sample breaks down before testing, the result will still be wrong. That brings in the next step, which is proving the sample stays stable long enough to be tested.

The Clock is Ticking: Ensuring Samples Do Not Spoil Before They Reach the Test Tube
Leave milk on a warm counter and it spoils. Blood samples with fragile drugs can do the same. If a sample degrades on the way from the clinic to the lab, even the best instrument cannot give the right result. A lab result depends on the quality of the sample. That is why scientists do stability testing of biological samples to show the drug has not broken down before measurement starts.
To prove that, researchers build stress tests into bioanalytical method development and validation. They expose samples to common conditions and check whether the drug survives:
- Bench-top stability: The drug stays intact while the sample sits at room temperature.
- Freeze-thaw cycles: The drug survives repeated freezing and thawing during transport and testing.
- Long-term storage: The sample stays stable in deep-freeze storage over time.
Passing these tests shows the robustness of quantitative mass spectrometry, the tool the lab uses like a magnifying glass to read the sample even after stress. Once the sample can survive handling and storage, the lab still has to show that the whole process meets legal and global standards.

Global Rules for Safety: How International Standards Like ICH M10 Protect Every Patient
If every country used a different ruler, working together would be hard. Drug testing used to face a similar problem because health agencies in different places had slightly different rules. The ICH M10 global harmonization guidelines help fix that. When labs use one shared rulebook, they avoid repeat work and support safer, more affordable drug development.
These rules also support honest science. Just as a restaurant needs to pass health checks, labs need GLP compliance in drug development. Good Laboratory Practice, or GLP, is a sign that the work was done with care and documented properly. By following the official guidance for industry bioanalytical method validation, labs create a record that shows each sample was handled accurately and ethically.
Science also changes over time. Labs may update equipment or move to a new building. That is where full and partial validation come in. Full validation builds the proof for a brand-new method. Partial validation checks an existing method after a smaller change. Together, they let labs adapt without dropping standards.

How Scispot Helps Teams Manage Bioanalytical Validation Work
Scispot is a strong fit for teams working under Bioanalytical Method Validation Guidance for Industry because it helps bring method development, validation, sample tracking, instrument data, SOPs, and review workflows into one system. Instead of splitting work across spreadsheets, shared drives, and disconnected tools, labs can keep calibration data, validation runs, stability studies, audit trails, and approvals in a single place. That makes it easier to stay organized, keep records inspection-ready, and trace every result back to its source. For bioanalytical teams that need accuracy, control, and clean documentation without adding more operational mess, Scispot is a practical digital layer to build on.
The Invisible Safety Net: Why You Can Trust the Data Behind Your Prescription
A simple lab result rests on a lot of proof. Turning a messy blood sample into a number doctors can trust takes a strong analytical method. By following strict bioanalytical method validation guidance for industry, scientists make sure the data used to approve a drug reflects what is really there.
Before a method is trusted, it has to pass this 5-point validation checklist:
- Accuracy: Measuring the true amount of the drug.
- Precision: Getting the same result each time the method runs.
- Selectivity: Measuring the drug without getting distracted by background noise.
- Sensitivity: Detecting even very small amounts of the drug.
- Stability: Showing the sample stays intact before testing.
This method validation process supports the safety of the healthcare system in ways most people never see. The next time you read a drug label and notice an exact dose, remember there is lab proof behind that number. That trust comes from methods that were tested hard, documented well, and shown to work.





.webp)
.png)

























.webp)
.webp)



