JPM 2026 week surfaced a common theme: building cursor/lovable/co-pilot for scientists. Here's how we're approaching this at Scispot.
Scispot is a system of record with an agentic layer for high-throughput, speed-conscious labs. Our customers range from R&D to well-regulated CLIA/CAP, GxP labs. We started as an alt-ELN, alt-LIMS, and over the last couple of years have been helping customers achieve sample-centric workflow outcomes using agents.
When we talk about "Cursor for life science," we're not talking about another chat window.
For pharma and serious platform biotech, it means three concrete capabilities:
1. Deep context
Agents operate on the same entities the business cares about: studies, samples, assays, runs,QC events, inventories, sites, and customers—not free-floating text.
2. Actionable tools
Agents don't just suggest; they invoke real operations like create_sample, register_manifest, assign_plate_well, link_to_experiment, attach_result, and issue_report.
3. Closed feedback loop with scientists and QA
Domain experts can correct the agent in natural language, and those corrections feed back into policies and patterns—without leaving their core workflow.
In Scispot, this becomes:
• Agents that understand your lab OS schema (samples, experiments, runs, QC, inventory, customers, vendors)
• A tightly controlled toolbox of operations they're allowed to call
• A review surface that shows exactly which records and fields will change before anything touches the system of record
Core principle: Agents propose. Humans dispose.
How aggressive those proposals can be is where non-regulated R&D and highly regulated environments diverge.

Architecture: The Four-Layer Stack
The four-layer architecture makes safe, high-speed agent operations possible in regulated environments. Scispot Agents orchestrate work around the system of record, not as a model embedded inside it.
Think of Scispot as two distinct but connected planes:
1. System of Record (SoR)
• The golden source for samples, experiments, results, inventory, approvals
• Fully audited, permissioned, and integrated with QMS, reporting, and downstream analytics
• Subject to validation and regulatory scrutiny
2. Agent Layer
• A reasoning and orchestration sandbox that can read from the SoR with role-based access and human approvals, apply policies, and propose changes
• Cannot directly mutate the SoR without going through explicit policy gates and/or human approval
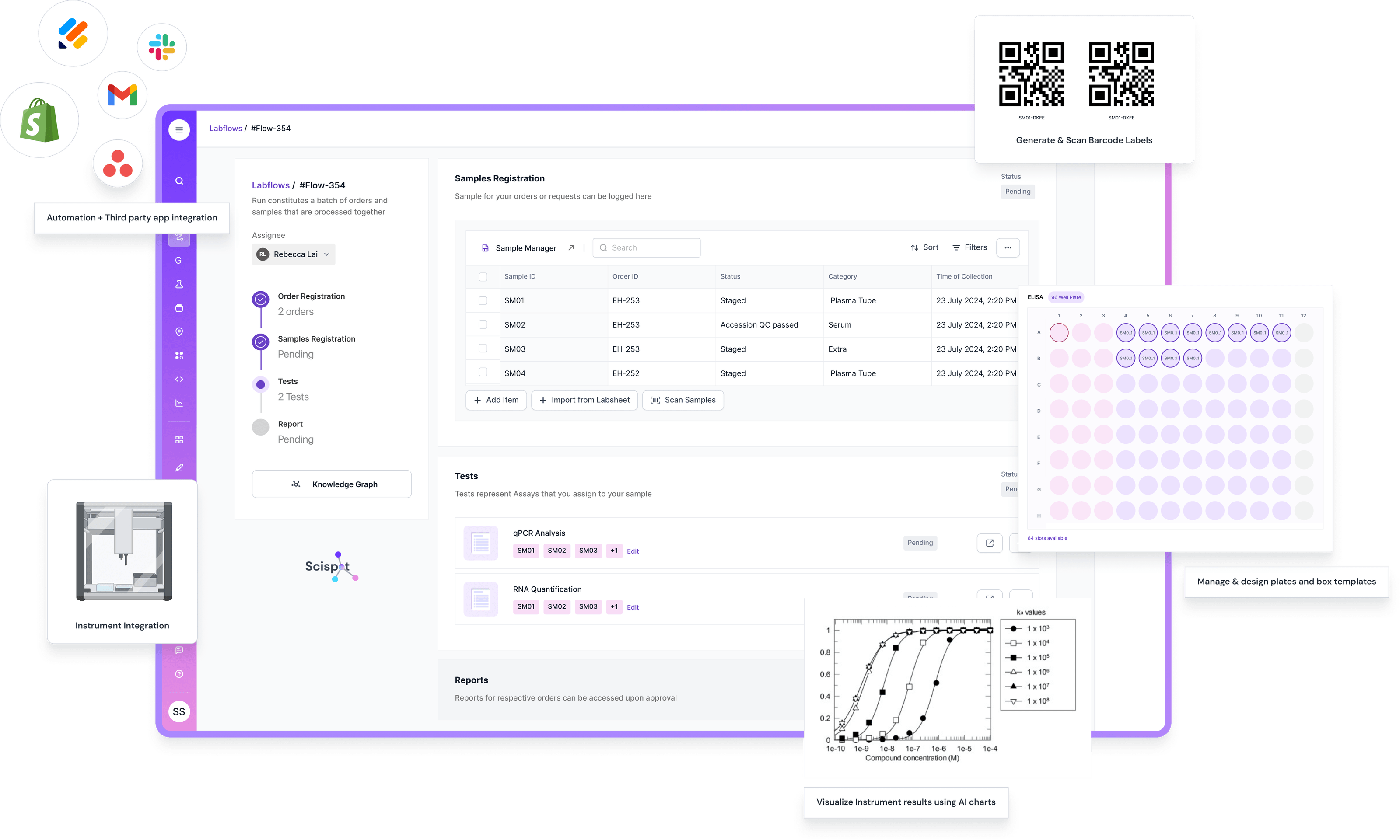
Workflow Example 1: Enterprise Sample-Intake
1. A manifest arrives (from a CRO, site, customer, or internal system) and lands in Scispot.
2. The agent parses the file, normalizes units/IDs/naming, cross-references
projects/studies/accounts in the SoR, proposes new samples/containers/plate layouts/links, and flags anomalies (missing consent, impossible volumes, protocol mismatches).
3. The agent emits a draft intake version with new records to create, existing records to update, and rationale for non-obvious decisions.
4. A reviewer sees a diff view: "Here's the exact delta between current state and the proposed state."
5. After approval, Scispot applies the diff to the SoR with version increments, full attribution(agent, model, human approver), and time-stamped, immutable audit logs.
From an enterprise perspective, this makes the agent feel less like an opaque model and more like a very fast, very consistent digital operator submitting structured change requests.
Workflow Example 2: Non-Regulated R&D—Turning Up the Autonomy Dial
In non-regulated R&D groups, the mandate is clear: accelerate cycle time, remove manual digital work, free scientists to focus on design and interpretation.
Here, Scispot Agents can operate with more autonomy, governed by risk-based policies:
Risk-tiered autonomy:
• Low-risk actions: Draft sample creation from manifests, tagging experiments, suggested plate layouts—often auto-applied with activity logging only
• Medium-risk actions: Updating sample metadata, rearranging inventory locations, assigning samples to runs—go through an in-context, one-click approval step
• High-risk actions: Deleting data, changing released results, regenerating formal reports—always require explicit human approval and justification
Workflow Example 3: Regulated Labs & Big Pharma—Plan → Review → Apply
In CLIA/CAP, GxP, and production environments, the calculus changes. The system of record is part of your validated state. Any change can have downstream regulatory, safety, and patient impact.

Scispot's model for regulated settings:
1. Plan (Agent)
The agent assembles a formal change plan with exact records to create or modify, fields, values, and reasons for each change, references to SOPs, runs, or prior states when relevant.
2. Review & Redline (Human)
Role-specific reviewers (QA, lab directors, study leads) see a structured diff showing old → new values, origin of the proposal (agent + model + context), and any policy flags. They can annotate, adjust, or reject portions of the plan.
3. Approve & Apply (System)
E-signature flows and access control enforce who can approve what. Only after approval dochanges propagate into the validated SoR. The full plan, discussion, and final diff become partof the permanent audit trail.
Crucially, because the agent sits outside the SoR:
• You validate the SoR and its APIs once
• You can iterate on agents, prompts, and policies with far more agility
• Model upgrades don't force you to re-validate the entire database
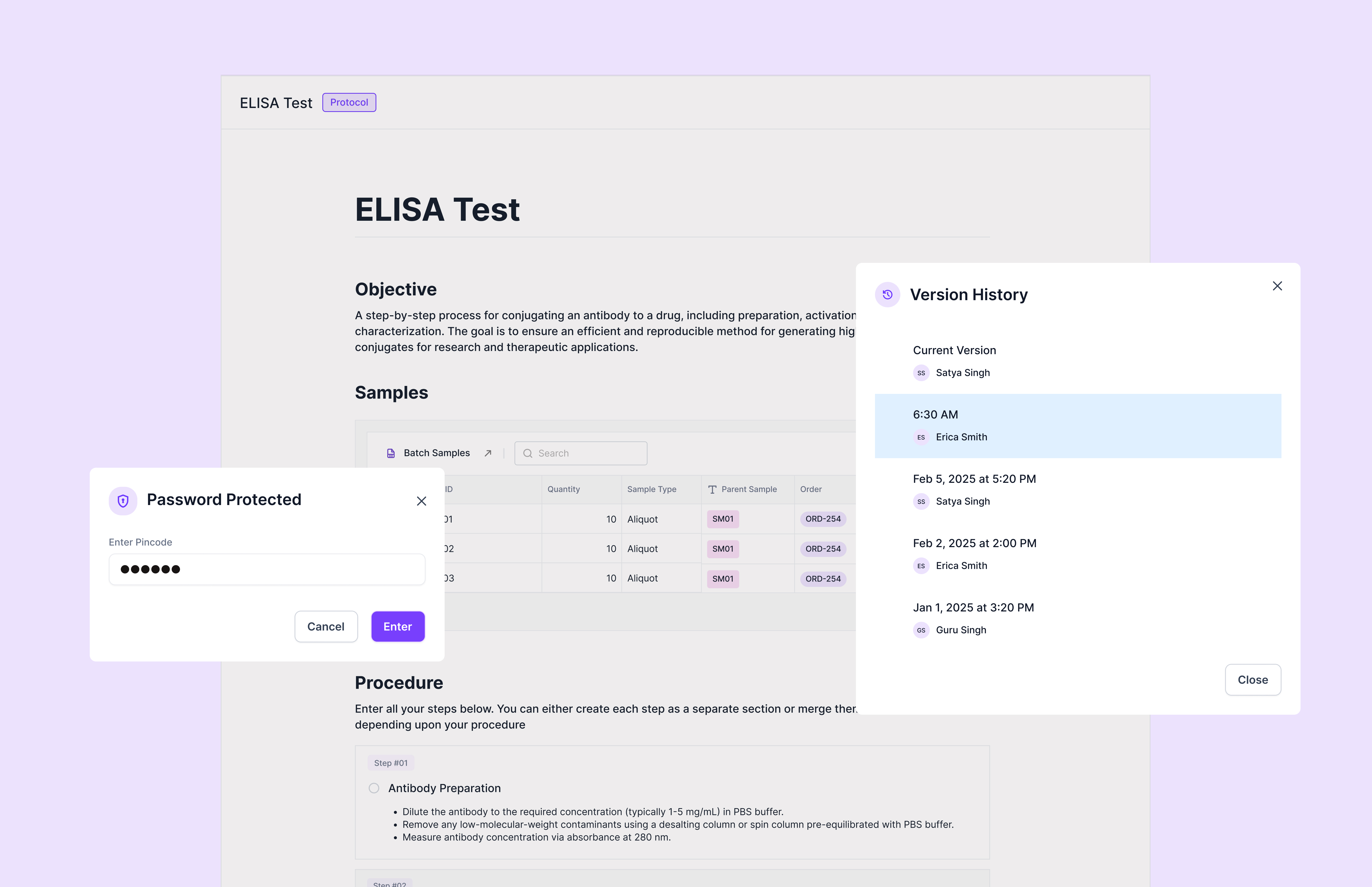
Versioning: The Trust Primitive
Any time AI can change critical lab data, leadership and regulators ask: "How do we prevent silent errors?" and "If something goes wrong, can we reconstruct what happened?"
Versioning is the core primitive that answers both.
In Scispot:
• Every agent-driven change corresponds to a version transition of specific objects
• You see, per object and per field: previous value → new value, which agent + which model proposed it, which human approved it, when, and under which policy
• You can time-travel and diff states to reconstruct how a sample, experiment, or report evolved
For regulated customers, this is a regulatory requirement. For investors and pharma leaders, it's the difference between "cool demo" and "production-grade platform that can survive an inspection."
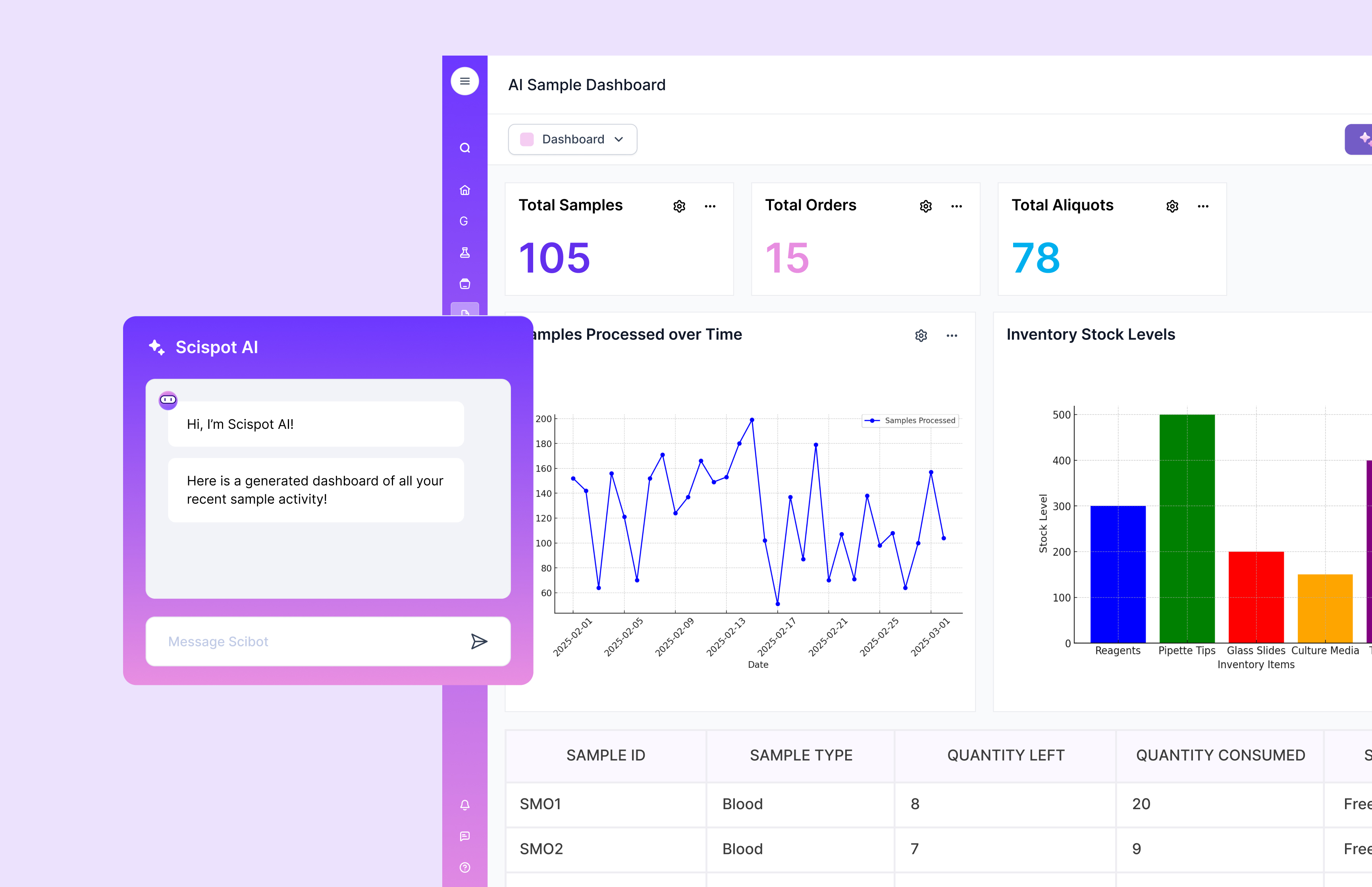
Human-in-the-Loop as Product, Not Slogan
If you simply prompt for approval on everything, two failure modes appear: scientists rubber-stamp approvals to get on with real work, or the organization disables the agent layer and reverts to manual workflows.
Scispot treats human-in-the-loop as a first-class product problem:
• Summarized change plans: Hundreds of low-level operations grouped into a handful of business-level changes. Details are expandable, not in your face.
• Inline corrections: Adjusting the plan is as fast as editing a spreadsheet cell; no context switching.
• Policy-aware prompts: The agent only escalates when it crosses defined risk or ambiguity thresholds; the rest is quietly logged and traceable.
• Role-specific views: Bench scientists, study leads, QA, and IT each see the appropriate level of abstraction and control.

This is what makes agents sustainable inside large organizations: you get leverage from humans in the loop, not drag.
Why This Matters
The next wave of value in life sciences won't come from standalone AI demos; it will come from deeply integrated agents that safely orchestrate work across lab and data systems.
Scispot's thesis:
The winning platforms will be those that become the control plane for lab agents—close enough to core data to be useful, but architected so that safety, compliance, and iteration speed can coexist.
By separating the agent layer from the validated system of record, and by grounding everything in versioned, reviewable change plans, Scispot can:
• Let non-regulated teams push autonomy and speed• Let regulated operations adopt agents within existing quality and validation frameworks• Give CIOs, Heads of R&D, and Quality leaders a credible path to "AI-first" labs without compromising on governance
If you're looking at where durable value will accrue in AI for life sciences, we believe it's here. But you have to build your harness layer along with human validation to control the agentic workflows while keeping the immutability of your System of Record.




%20(2).webp)

























.webp)
.webp)



