Can you explain the basic principles behind molecular diagnostics?
When a detective arrives at a crime scene, they do not just look at the broken window. They look for fingerprints. Molecular diagnostics works in a similar way. Instead of only looking at symptoms like a cough or fever, doctors look for small biological clues inside the body.
Every disease leaves signs behind. Tests that analyze DNA and RNA can show what is making someone sick, often before major symptoms appear. Viruses and bacteria carry unique genetic sequences, so this method is more precise than simple observation. It helps doctors identify the real cause of illness and choose treatment with more confidence.
This is also the idea behind precision medicine. If doctors can spot the exact genetic evidence of disease early, they can move away from guesswork and make care more personal and accurate.
Your body’s blueprint: how DNA and RNA help reveal disease
A simple way to think about this is to picture your body as a construction site. DNA is the master instruction manual kept in the main office. Since that original set of instructions stays protected, cells make a temporary copy called RNA to carry directions to the rest of the site.
Sometimes small errors show up in that copy. One changed letter in a genetic sequence may look minor, but it can lead to serious illness. Scientists found that these changes can act as early warning signs, sometimes long before a person feels sick.
Modern molecular diagnostics tries to catch those mistakes early. Instead of waiting for symptoms to become obvious, doctors examine these biological instructions for signs of trouble. That gives them a more direct way to find disease and guide treatment.
The hard part is spotting one wrong letter in a huge amount of genetic code. Modern machines do that by searching for specific genetic barcodes instead of waiting for germs to grow.

Why looking for barcodes is faster than waiting for germs to grow
For years, diagnosing an infection often meant taking a sample and waiting for it to grow in a petri dish. That process can take days. Molecular testing works differently. It looks for the genetic blueprint of the germ right away.
Scientists search for biomarkers that act like genetic barcodes. Each virus or bacterium has a distinct sequence in its DNA or RNA. When a test finds that sequence, doctors can detect the pathogen much faster, often in hours instead of days.
This also improves the odds of getting the diagnosis right the first time. That is where sensitivity and specificity matter. High sensitivity helps detect even very small traces of disease, which lowers the risk of false negatives. High specificity helps the test avoid mistaking one harmless organism for a harmful one.
When doctors know what is causing the illness, they can choose the right treatment sooner. To get there, scientists first have to pull the genetic material out of a messy human sample.

From swab to result: how molecular extraction works
A swab sample is not clean. It contains mucus, human cells, harmless bacteria, and other material. To find the genetic signal of disease, scientists have to separate it from everything else. That prep step isolates the DNA or RNA needed for testing, so the result is not distorted by the rest of the sample.
The process usually includes a few main steps:
- Breaking open cells (lysis): Chemicals break open cells and release the DNA or RNA inside.
- Removing proteins and fats: Enzymes and other reagents clear away unwanted material.
- Washing the genetic material: The sample is cleaned to remove leftover contaminants.
- Releasing the purified sample: The final DNA or RNA is placed in liquid and prepared for testing.
Even after cleanup, the sample can still be damaged by contamination. Molecular tests are very sensitive, so even a tiny amount of outside DNA can affect the result. That is why lab staff work in clean airflow cabinets and use protective gear.
Once the sample is purified, the next issue is quantity. There is often too little genetic material to read directly. That is why scientists use PCR to make many more copies.

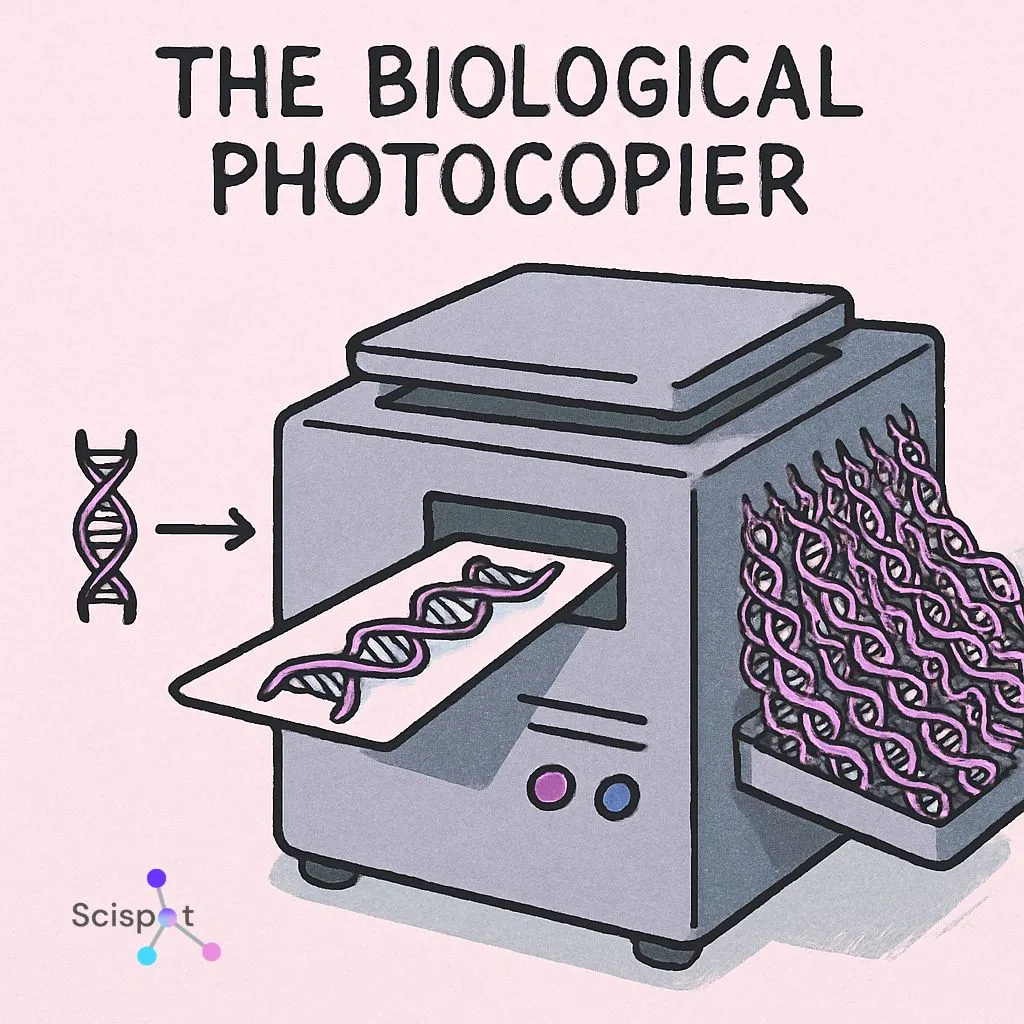
How PCR works like a biological photocopier
Think about finding one tiny fingerprint at a crime scene. It is there, but it is too small to study on its own. PCR, or Polymerase Chain Reaction, solves that problem by making many copies of a small piece of genetic material until machines can detect it.
Before copying starts, scientists make sure they target only the disease-related DNA or RNA, not the patient’s normal genetic material. They do this with primers. Primers act like very specific search terms. They are designed to find and attach to the exact sequence linked to the virus, bacterium, or mutation being tested.
Once the target is found, the machine runs cycles of heating and cooling. Each cycle doubles the amount of target DNA. After enough cycles, one small fragment becomes billions of copies.
That is what makes PCR so useful. It can detect an infection even when only a tiny amount of genetic material is present. It works especially well when doctors already know what they are looking for. But if the cause of illness is still unclear, they may need a broader method.
Why next-generation sequencing changed the field
PCR is great when there is a clear suspect. But sometimes doctors do not know what they are looking for. In those cases, next-generation sequencing, or NGS, gives a much wider view.
Instead of testing for one target, NGS reads large amounts of genetic material at once. Machines break the sample into pieces and read many fragments in parallel. That gives doctors a much fuller picture of what is happening.
This produces a huge amount of data, so computers play a major role. Bioinformatics software sorts through billions of letters and compares them with known reference sequences. That helps scientists find mutations or patterns linked to disease.
This broader approach is especially useful in complex cases, including rare diseases and many cancers. It gives doctors more information and supports more tailored care.
How scientists actually “see” DNA
DNA is too small to see directly, so scientists use markers and signals to detect it. One example is fluorescent in situ hybridization, or FISH. In this method, fluorescent labels attach to specific DNA sequences. Under a microscope, those labeled areas light up, which helps doctors spot changes inside cells.

Labs use several methods to turn invisible molecules into readable results:
- Fluorescent signals (FISH): Used to highlight specific genetic targets in cells.
- Color or signal changes (real-time PCR): Used to measure genetic material during amplification.
- Digital signals (digital PCR): Used for very precise counting of target DNA molecules.
Digital PCR goes a step further than standard real-time PCR. It separates DNA strands into many tiny partitions and reads each one as present or absent. That gives a more exact count and can detect very small amounts of disease-related material.
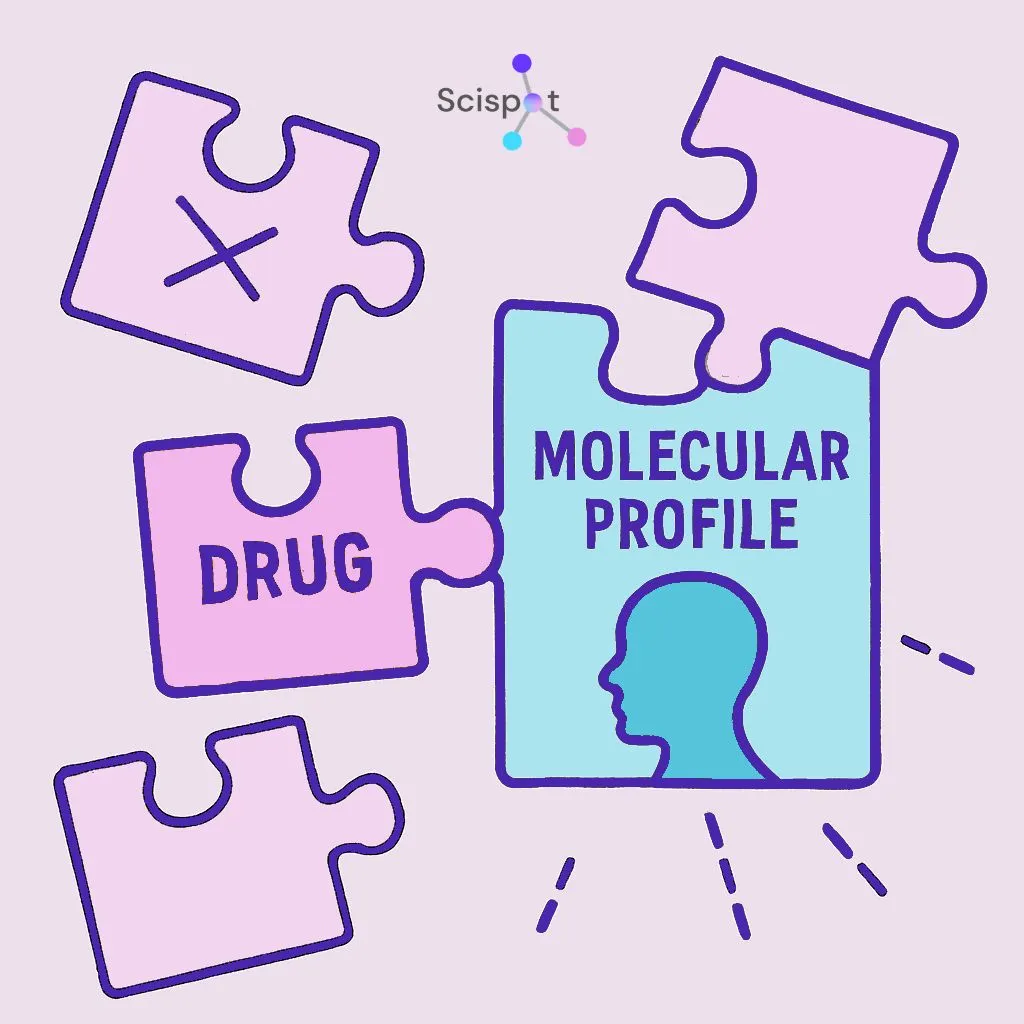
Why precision medicine matters
One of the biggest benefits of molecular diagnostics is that it helps doctors match treatment to the patient more accurately. Instead of relying on trial and error, they can use the biological details of a disease to guide care.
If a test shows the exact mutation, pathogen, or marker involved, doctors can choose a treatment that fits that specific case. That can reduce time lost on treatments that are unlikely to work and may spare patients unnecessary side effects.
This approach is also becoming more available at the point of care. Some molecular testing devices now sit in clinics or doctors’ offices instead of distant labs. That means patients can sometimes get answers much faster and begin treatment sooner.
How this helps you make better healthcare decisions
Understanding the basic ideas behind molecular diagnostics makes the process less abstract. Instead of waiting for symptoms to get worse or for organisms to grow in culture, doctors can now look for genetic evidence much earlier. That has changed how many diseases are diagnosed and treated.
It also helps patients ask better questions. If you are dealing with a new diagnosis, useful questions may include:
- Is there a molecular test for this condition?
- Will this test help guide treatment?
- How quickly can we get the results?
Molecular diagnostics gives doctors more direct evidence about what is happening inside the body. That is a big part of why it now sits at the center of precision medicine. The more care is guided by real biological data, the better the chance of choosing the right path early.





.webp)
.png)

























.webp)
.webp)



